 Theoretical studies have often been employed to guide experiments and to identify unknown polynitrogens. These successes led to an increasing interest in polynitrogen compounds and to the search of other stable polynitrongen forms in recent years. Around the same time, the N 5 − anion 13, neutral N 4 molecule 14 have also been isolated experimentally. Subsequently, thermally more stable N 5 + SbF 6 − and N 5 + Sb 2F 11 − have been synthesized 12. reported a successful synthesis of marginally stable N 5 + AsF 6 − crystal, which contains the second polynitrogen ion, the N 5 + cation 11.  
Producing new polynitrogens involves substantial experimental difficulties due to the metastability of the products. Before 1999, the azide anion (N 3 −), which exists in metal azides (i.e., NaN 3, Pb(N 3) 2), was the only known polynitrogen ion. The realization of polynitrogens has been actively experimented in nitrides, due to the fact that the charged nitrogen species in such materials often show improved kinetic stability over pure polynitrogens. 
Solely single-bonded polynitrogen, for example, has an estimated energy capacity of 4.6 eV/mol, about three times that of the most powerful energetic materials known today 10. This is possible if one can transform the diatomic N 2 molecules into single- or double-bonded polynitrogen, utilizing the large energy difference between the single and double/triple bonds 1, 2, 3, 4, 5, 6, 7, 8, 9. Nitrogen, which is abundant in nature, may be sought as a high-energy-density material (HEDM). The pursuit for new and efficient energy source has always been a focus of scientific research. Results of the present study suggest that it is possible to obtain energetic polynitrogens in main-group nitrides under high pressure. To our best knowledge, this is the first time a charged N 4 species being reported. We also identified a very interesting CsN crystal that contains novel N 4 4− anion. This structure contains a substantially high content of single N-N bonds that exceeds the previously known nitrogen chains in pure forms and also exhibit metastability at ambient conditions. Polymeric chains of nitrogen were found in the high-pressure C2/ c phase of CsN 2. Nitrogen species in the predicted structures have various structural forms ranging from single atom (N) to highly endothermic molecules (N 2, N 3, N 4, N 5, N 6) and chains (N ∞). Along with the CsN 3, we identified five new stoichiometric compounds (Cs 3N, Cs 2N, CsN, CsN 2 and CsN 5) with interesting structures that may be experimentally synthesizable at modest pressures ( i.e., less than 50 GPa). Using a structure search method based on the CALYPSO methodology, we investigated the stable stoichiometries and structures of cesium polynitrides at high pressures. 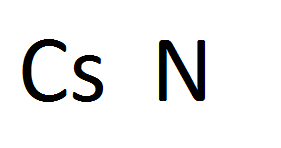
Cesium Nitrate Powder SpecificationĬesium Nitrate is used in pyrotechnic ingredients as colorants and oxidants, such as in bait bombs and flares.New polynitrides containing metastable forms of nitrogen are actively investigated as potential high-energy-density materials. Nanoscale elemental powders and suspensions, as alternative high surface area forms, may be considered. Ultra-high purity and high purity compositions improve both optical quality and usefulness as scientific standards. Cesium Nitrate is widely applied in glass, electronic and chemical industries.Ĭesium Nitrate is generally immediately available in most volumes. It’s the raw material of various flameproof glass and catalysts. Cesium Nitrate, or Caesium Nitrate, is a compound that is highly soluble in water. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
